HCLS/F2F/DrugSafety
Appearance
HCLS Drug Safety&Efficacy DSE main page
- Scenario collection and discussion around Clinical Trails (CT) data collection/aggregation and translational analysis
- Making SDTM data "Recombinable" across studies (Kerstin Forsberg)
- Translational Science Scenario (Bo Andersson)
- Scenario...
- Scenario...
- Relevance and Strategy for mapping CDISC SDTM into RDF-OWL frame
- Study Data Tabulation Model (SDTM)
- SDTM elements or functionality to focus on
- Multiple Data Tables (Sets) from studies
- Genomic and Biomarker data in studies and submissions (trend shown below)
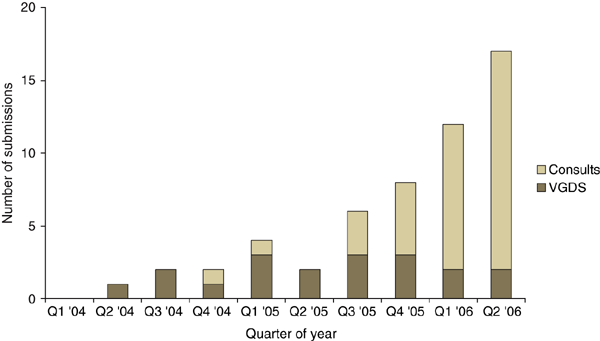 http://www.nature.com/nbt/journal/v24/n9/full/nbt0906-1105.html
http://www.nature.com/nbt/journal/v24/n9/full/nbt0906-1105.html
1.#3 Controlled vocabularies (URI nodes) used in SDTM obeservational elements
- Discussion (around scenarios?) about defining annotation layers around clinical information systems (JANUS?)
- Basic Janus model
- Necessary Components needed for supporting Translational Research
- Biomarker linking (retrospective and follow-up studies)
- Mechanism (Pathway) linking: latest knowledge automatically updated
- Target knowledge assembly
- Comprehensive linking of Toxicity information
- Access Control and security
- HIPAA/21Part11 issues to be identified